 
The temperature ( T) speeds up a conversion exponentially, because the productivity is proportional to (T ø. The Temperature impacts the Reaction Rate, Energy Efficiency, Heat-Transfer Rate and the Equilibrium Constant. The main factors that influence productivity are shown in the highlighted table below. 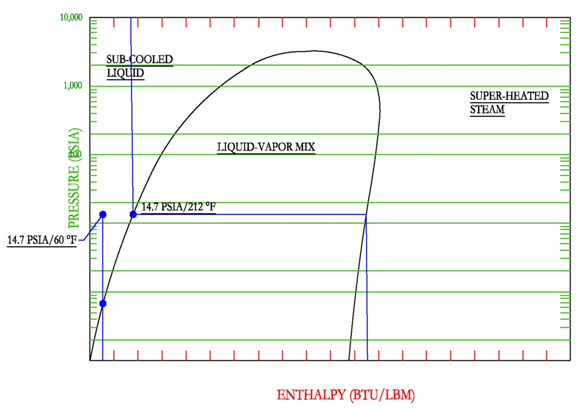
The commonly employed unit for E, is kJ/mol for chemical reactions. The symbols R and E below represent the Universal Gas Constant and a generic activation barrier respectively. Productivity and production-amount activity are interchangeable terminologies. Better productivity generally leads to better energy efficiency. Improvements in productivity are achieved through improved operation-research optimization methods, good internal business practices and best technical conditions that keep up with the latest technical improvements in thermal and materials availability. Overall the productivity of a Commercial Chemical Process is defined as the amount of product made by an industrial chemical reaction plant (units of tons/year). The rate of reaction involves the equilibrium constant and a few other kinetic ‘processing variable’ discussed below. Process variables are initial temperature, pressure, concentration of reactants and other pertinent conditions like the quality of insulation, feed back control-ability, good sensors and like. For chemical reactions the productivity can be considerably influenced by changes in the imposed process conditions. K is is a thermodynamic property thus implying some strict boundaries for the possibilities to influence this number without changing other variables like temperature. The direction of any reaction is influenced by the Equilibrium Constant, K (defined below). 
The four bullets below show that the productivity can be mainly enhanced by an increase in the Temperature (T), only somewhat by Pressure (P) and sometimes with the use of a good Catalyst. tons per year) may be enhanced by tweaking the process variables like temperature, the pressure or enhancing heat transfer rates where applicable. 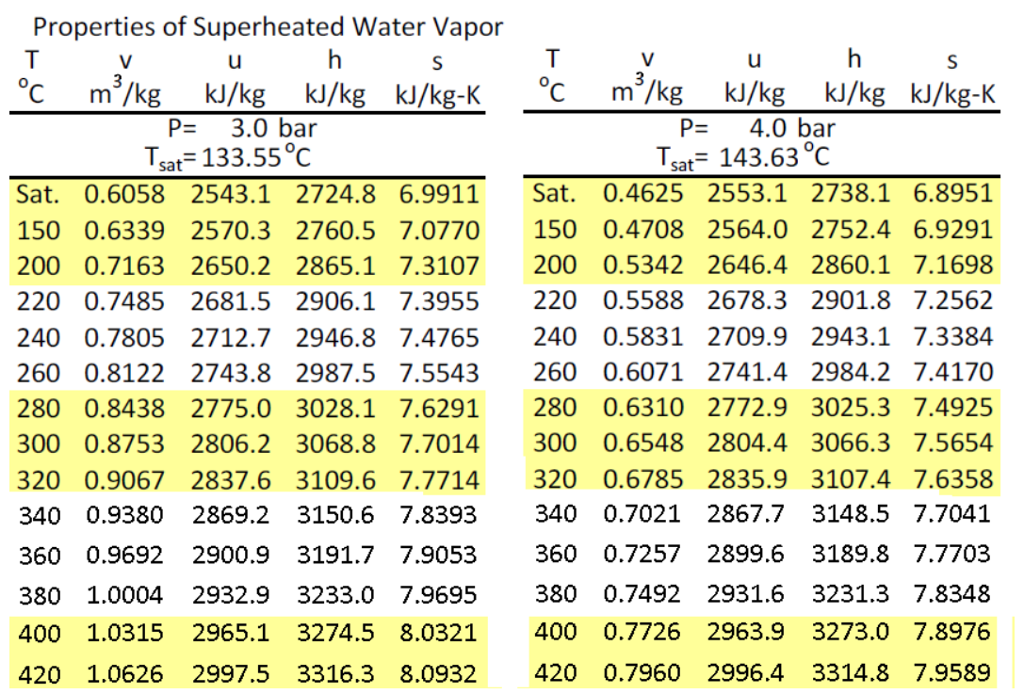
Below, we focus mainly on the the technical variables that influence this productivity (sometimes also referred to as activity in the productivity literature).Īs a rule of thumb, an increase in the productivity of an heat-enabled favorable reaction, measured in throughput increase per unit time (e.g. The term industrial-productivity, encompasses not only the chemical kinetic rate but also aspects of the long-term product life-cycle and other important economic factors that define the reactant and other supply processes (see also innovation constants). To enable an increase in the productivity of a chemical or materials processing method, the ojective is to increase the conversion rate of reactants to products. Graphs Water density as function of temperature at 1.01325Bar(A) Water specific isobaric heat capacity as function of temperature at 1.01325Bar(A) Water specific isochoric heat capacity as function of temperature at 1.01325Bar(A) Water specific enthalpy as function of temperature at 1.01325Bar(A) Water specific entropy as function of temperature at 1.01325Bar(A) Water internal energy as function of temperature at 1.01325Bar(A) Water dynamic viscosity as function of temperature at 1.01325Bar(A) Water kinematic viscosity as function of temperature at 1.More Detailed Answer: Please read on….for high temperature steam and the novel open-ionic products. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
